What is lithium ion battery?
The lithium ion battery has a system that is uniquely different than other batteries similar to it (Bellis, 2015). The lithium ion battery has used non-aqueous electrolyte, which is composed from organic liquids and salts of lithium to provide ionic conductivity (Bellis, 2015). This system has much higher voltages cell than the other aqueous systems (Bellis, 2015). Although they require more difficult assembly, as it must be done in a nearly perfectly dry atmosphere (Bellis, 2015). The lithium ion battery cells can operate with wider potentials, due to the exclusion of water, the evolution of hydrogen and oxygen gases is eliminated (Bellis, 2015).
further explanation on the lithium ion battery
how does it work?
Electrochemistry is when you are generating electricity using chemical reactions. The chemical nature of reactions causes the negative and positive charges to react to make energy in the form of electricity. The lithium battery is similar to all other types of batteries in the way it works (Templeton, 2014). Lithium ions move through the electrolytes (liquid conductor that moves electrons) from the positive and negative electrodes (Templeton, 2014).
In a completely discharged battery, the mobile lithium ions will be connected to the positive electrode (Templeton, 2014). The lithium ion batteries have chemical properties, which allow them to jump to the positive electrode material even as they lack electrons (Templeton, 2014). We can then give them electrons by thrusting electricity into the system, which is called recharging (Templeton, 2014). They then will detach from the positive electrode and transfer back to the negative one (Templeton, 2014). When they all lined up on the negative electrode, with high energy electrons, we than call the battery “charged” (Templeton, 2014).
When the battery is in the state of being charged, where all the electrons are attached to the negative electrode, we can now make the opposite (Templeton, 2014). We can open a path for electrons that are attached to the negative electrode to travel down their charge slope to the positive side of the battery (Templeton, 2014). This takes away the electrons from the negative electrode and makes them again positive (Li+), causing them naturally to transfer all the way back (Templeton, 2014). We can use the electron flow of the negative to positive, to power everything from pacemakers to electric cars, and it all ultimately comes down to the back and forth movements of ions (Templeton, 2014). However, recently the scientists discovered why too many back and forth reactions cause a battery to slowly die (Templeton, 2014).
In Li-ion batteries, lithium ions move from the anode to cathode during discharge, and from cathode to anode when charging (Nexeon, 2015). The type of material that is used to act as the anode or cathode changes many factors of the battery such as capacity, battery length, and more(Nexeon, 2015).
An issue regarding the Li-ion batteries is their lifespan and the amount of energy they can have (Nexeon, 2015). Currently there is search of materials that are able to hold high-energy amount and also be able to have a high lifespan, which makes the battery more efficient (Nexeon, 2015). The use of new cathode, anode, and electrolytes will significantly improve the performance of Li-ion batteries (Nexeon, 2015).
FURTHER EXPLANATION OF HOW LITHIUM ION BATTERIES WORK
Chemical reactions in the lithium ion batteries
Overall reaction on a Li-ion cell: C + LiCoO2 ↔ LiC6 + Li0.5CoO2 (Nexeon, 2015)
At the cathode: LiCoO2 – Li+ – e- ↔ Li0.5CoO2 ⇒ 143 mAh/g
(Nexeon, 2015)
At the anode: 6C + Li+ + e- ↔ LiC6 ⇒ 372 mAh/g
(Nexeon, 2015)
At the cathode: LiCoO2 – Li+ – e- ↔ Li0.5CoO2 ⇒ 143 mAh/g
(Nexeon, 2015)
At the anode: 6C + Li+ + e- ↔ LiC6 ⇒ 372 mAh/g
(Nexeon, 2015)
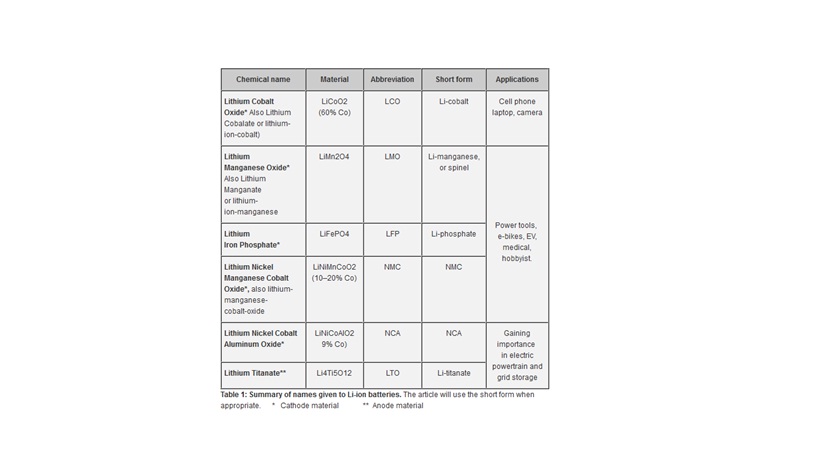
Types of lithium ion batteries
Advantages and disadvantages
Advantages
According to the Isidor Buchmann there are 5 advantages of lithium ion batteries
1. High energy density – Li-ion batteries have the potential.
2. Does not need prolonged priming when new. One regular charge is all that's needed.
3. Comparatively low self-discharge - self-discharge is almost 50% less than nickel-based batteries.
4. Low Maintenance - no periodic discharge is needed; there is no memory.
5. Special cells in the battery are able to provide high current to tools such as power tools
(Buchmann, 2015).
1. High energy density – Li-ion batteries have the potential.
2. Does not need prolonged priming when new. One regular charge is all that's needed.
3. Comparatively low self-discharge - self-discharge is almost 50% less than nickel-based batteries.
4. Low Maintenance - no periodic discharge is needed; there is no memory.
5. Special cells in the battery are able to provide high current to tools such as power tools
(Buchmann, 2015).
disadvantages
According to the Isidor Buchmann there are 5 disadvantages of lithium ion batteries
1. Their high voltage and current makes them require a protection circuit to keep these factors within safe limits.
2. Ages quickly. Even if it is not in use it ages quickly - storage in a cool place at 40% charge reduces the aging effect.
3. Transportation restrictions - shipment of larger quantities require regulatory control due to their hazards.
4. Li-ion batteries are expensive to manufacture - approximately 40 % higher in cost than nickel-cadmium batteries
5. The anode and cathode metals, as well as the materials for the electrolyte change due to the reactions and different external factors (Buchmann, 2015).
1. Their high voltage and current makes them require a protection circuit to keep these factors within safe limits.
2. Ages quickly. Even if it is not in use it ages quickly - storage in a cool place at 40% charge reduces the aging effect.
3. Transportation restrictions - shipment of larger quantities require regulatory control due to their hazards.
4. Li-ion batteries are expensive to manufacture - approximately 40 % higher in cost than nickel-cadmium batteries
5. The anode and cathode metals, as well as the materials for the electrolyte change due to the reactions and different external factors (Buchmann, 2015).